– The endoscopic AI differentiates gastric lesions as neoplastic or non-neoplastic –
TOKYO–(BUSINESS WIRE)–AI MEDICAL SERVICE INC. (hereinafter AIM), a medical start-up specializing in the development of diagnostic endoscopic AI, is excited to announce the first instance of Singaporean regulatory review and device registration for an AI-based endoscopic diagnostic support device that operates within the stomach (brand name: Endoscopic image diagnosis support software – gastroAI-model G). The regulatory review and device registration was conducted by Singapore’s Health Sciences Authority (hereinafter HSA) on February 19, 2024. The endoscopic AI (hereinafter gastric cancer differentiation AI) differentiates gastric lesions and judges them as either neoplastic or non-neoplastic in nature. In addition, AIM has confirmed that this is the first case of device registration in Singapore for an endoscopic support software that operates within the upper gastrointestinal tract.
Background of our efforts to complete regulatory review and device registration in Singapore
Gastric cancer is the fifth most common cancer in the world, with more than 1 million people contracting gastric cancer and approximately 770,000 people dying from the disease each year. Gastric cancer is characterized by a mortality rate that increases significantly with disease progression. However, gastric cancer is very often treatable if detected at an early stage; the 5-year relative survival rate is approximately 95% if detected at stage I, but less than 50% if detected at stage III or later. Nevertheless, early-stage gastric cancer is difficult to detect and is said to be missed in 4.5 to 25.8% of cases.
In countries with a high incidence of gastric cancer like Japan and South Korea, endoscopic gastric cancer screening is routinely performed nationwide. However, such screening is not a routine practice in Singapore and other countries, further complicating the early detection of gastric cancer.
Considering these circumstances, AIM has been conducting joint research on endoscopic AI with National University Hospital of Singapore (hereinafter NUH). AIM and NUH reported that endoscopic AI can support endoscopists’ ability to diagnose and potentially find gastric cancer at an early stage1. In addition, through communication with local physicians and activities at academic conferences, AIM has increased awareness regarding the value that endoscopic AI can provide to clinical practice in Singapore. Concurrent to that, AIM spent years working to complete regulatory review and device registration in Singapore.
About gastric cancer differentiation AI “Endoscopic image diagnosis support software – gastroAI-model Gâ€
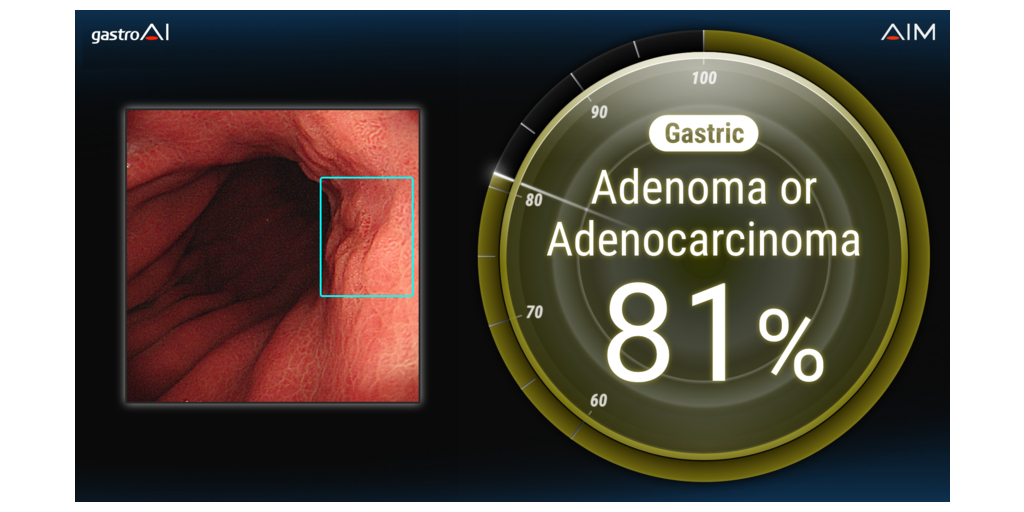
The gastric cancer differentiation AI, brand-named “Endoscopic image diagnosis support software – gastroAI-model G,†is a diagnosis support system for determining the neoplastic or non-neoplastic nature of gastric lesions. The system analyzes endoscopic still images acquired during endoscopic examination and provides near-instant feedback to the physician. The software indicates whether the candidate lesion featured in the image is likely neoplastic (“Adenoma or Adenocarcinomaâ€), likely non-neoplastic, or unlikely to be neoplastic (“Low Confidenceâ€)2. If the lesion in the image is neoplastic, the degree of visual similarity between it and historical neoplastic lesions (confidence level) is displayed, and a rectangle indicating the location of the lesion is superimposed onto the image.
Brand name | Endoscopic image diagnosis support software – gastroAI-model G |
Registration number | DE0508971 |
Future Developments
AIM has been conducting joint research with NUH, a top-ranked university in Asia. AIM will continue to develop AI products through joint research with NUH, expanding device functions and target organs in line with the challenges faced by patients practitioners in Singapore. In addition, utilizing regulatory approval in Singapore, AIM aims to obtain approval and develop our business in other ASEAN countries through “the ASEAN Medical Device Directive,†a common framework that aims to improve efficiency and simplify the application process regarding the registration of medical devices in ASEAN countries.
About AI Medical Service Inc.
AI Medical Service (AIM) is a Tokyo-based med-tech company established with the mission to “Save Lives All Over the World.†Japan leads the world in endoscopic diagnosis and treatment, providing firms and researchers with access to large amounts of high-quality data. AIM is the leading player in the field of endoscopic AI, engaging in joint research with more than 100 medical institutions. By bringing endoscopic AI to the real-world clinical setting as soon as possible, AIM aims to reduce the number of missed cancer diagnoses and save lives around the world.
About CEO of AIM, Dr. Tomohiro Tada
Tomohiro TADA, M.D., Ph.D.
Dr. Tomohiro TADA is the CEO of AI Medical Service Inc., the chairperson of Tada Tomohiro The Institute of Gastroenterology & Proctology as well as Visiting Lecturer, Department of Surgical Oncology, Graduate School of Medicine, the University of Tokyo Hospital.
Dr. TADA received his M.D. of school of medicine in 1996 and Ph.D. of department of surgery in 2005 from the University of Tokyo. He trained in Colorectal Surgery at the University of Tokyo Hospital.
Company: AI Medical Service Inc.
Address: Hareza Tower 11F, 1-18-1 Higashiikebukuro, Toshima-ku, Tokyo 170-0013, Japan
CEO: Tomohiro Tada
Founding: September 1, 2017
Business: Development of Endoscopic AI
1 Reference: So JBY et al. Gut. 2020. doi: 10.1136/gutjnl-2020-322065
Koh C, Tada T et al. JGH. 2023. doi: 10.1111/jgh.16274
2 This product classifies images into three categories: “neoplastic,†“non-neoplastic,†and “low confidence.†Among these, “non-neoplastic†and “low confidence†are collectively displayed as “Low Confidence.â€
Contacts
AI Medical Service Inc.
Manami Taniguchi
[email protected]